Our Treg suppression assay formats are adaptable, using natural Tregs isolated from PBMCs, or induced Tregs expanded and differentiated from naïve CD4+ T cells. Routinely, autologous PBMCs are used as the population to be suppressed, however, the system is open to modification to use pan-T cells, CD4+ or CD8+ T cells as required. Our expert immunologists work closely with you to design the most appropriate system to achieve your goals.
What are regulatory T Cells?
Tregs have the ability to suppress the function of other lymphocytes by releasing immunosuppressive cytokines such as IL-10 and TGFβ. These cytokines also act to promote polarisation of CD4+ T cells in the periphery to create more Tregs, acting as a positive feedback loop. Tregs can inhibit the activities of CD4+ and CD8+ effector T cells, Natural Killer (NK) cells, NKT cells and antigen presenting cells (APCs) through multiple mechanisms.
Tregs are defined by functional and phenotypic markers, most notably the expression of the transcription factor FoxP3 and high expression of CD25.
An imbalance in the number of Tregs is associated with various disease states. Too many Tregs can lead to immunosupression, creating an environment which favours the survival of tumour cells. Conversely, too few Tregs are associated with several autoimmune disorders as the immune system is left unchecked.
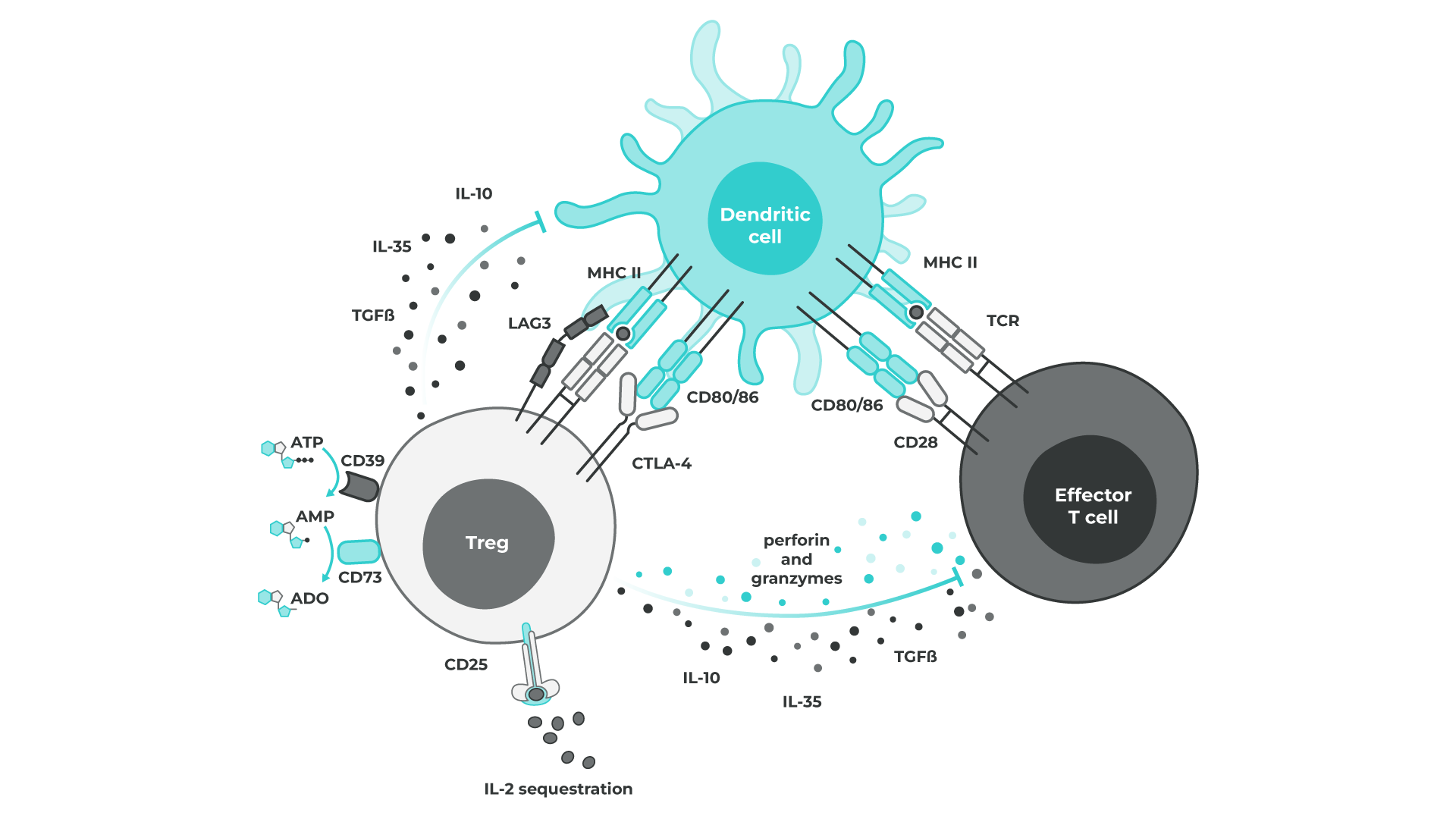
Figure 1: Tregs mediate suppression by multiple mechanisms regulating inflammatory processes, preventing auto-immunity and also contributing to poor tumour immunity. Tregs can act directly on responding effector T cells via immunosuppressive cytokines e.g. IL-10, IL-35, TGFbeta; metabolic interference e.g. production of adenosine (ADO) from ATP, sequestration of IL-2 and even cytolysis. These mechanisms can also impact antigen presenting cells such as dendritic cells, dampening their activation and ability to effectively stimulate adaptive immune responses.
What is a Treg suppression assay?
When combined in vitro, Tregs inhibit the proliferation and activation of T cells in response to recognition of specific antigen. Tregs are combined with autologous PBMCs (containing both responder CD4+ and CD8+ T cells as targets for suppression) and a source of T cell stimulation e.g. anti-CD3. Since natural Tregs exist in low numbers, this can limit assay size, however use of expanded iTregs allows larger scale assays to be performed.
Proliferation and activation of the responder T cells is determined in the presence and absence of Tregs so that the degree of suppression caused by the Tregs can be expressed in terms of inhibition of maximal responses. This can be coupled with cytokine release measurements (ELISA or multiplex cytokine analysis) and phenotyping for activation markers in the regulatory and responder populations to provide high content information to delve deeper into the mechanistic effects of a test therapeutic.

When to use Treg suppression assays
A number of therapeutics in development aim to target Treg function in an attempt to alleviate immuno-suppression. One proposed mechanism of action of the approved CTLA-4 targeting antibody, ipilimumab, is that it enhances antitumor immunity by inhibiting the immunosuppressive activity of regulatory T cells. Anti-CTLA4 binding to Tregs can simultaneously mark them for ADCC-mediated destruction.
Learn more about Antibody Analytics’ Treg suppression models
In our whitepaper, we describe how scientists from Antibody Analytics have established a Treg suppression assay workflow. The assay covers the isolation of natural Treg cells from peripheral blood mononuclear cells (PBMCs) and co-culture with anti-CD3 stimulated autologous PBMCs at different Treg to PBMC ratios to demonstrate functional suppression of the responder (conventional or Tconv) T cell population. This is coupled with Treg immunophenotyping, cytokine release and Treg-specific reversal of suppressive activity with a novel immunotherapy.




